

According to the US Food and Drug Administration (FDA), a ‘device recall’ is defined as, “when a manufacturer takes a correction or removal action to address an issue with the medical device that violates the FDA law”1.

Diabetes Mellitus (DM) is an endocrine metabolic disorder, characterized by elevated blood glucose level. DM is sub-classified into following categories.
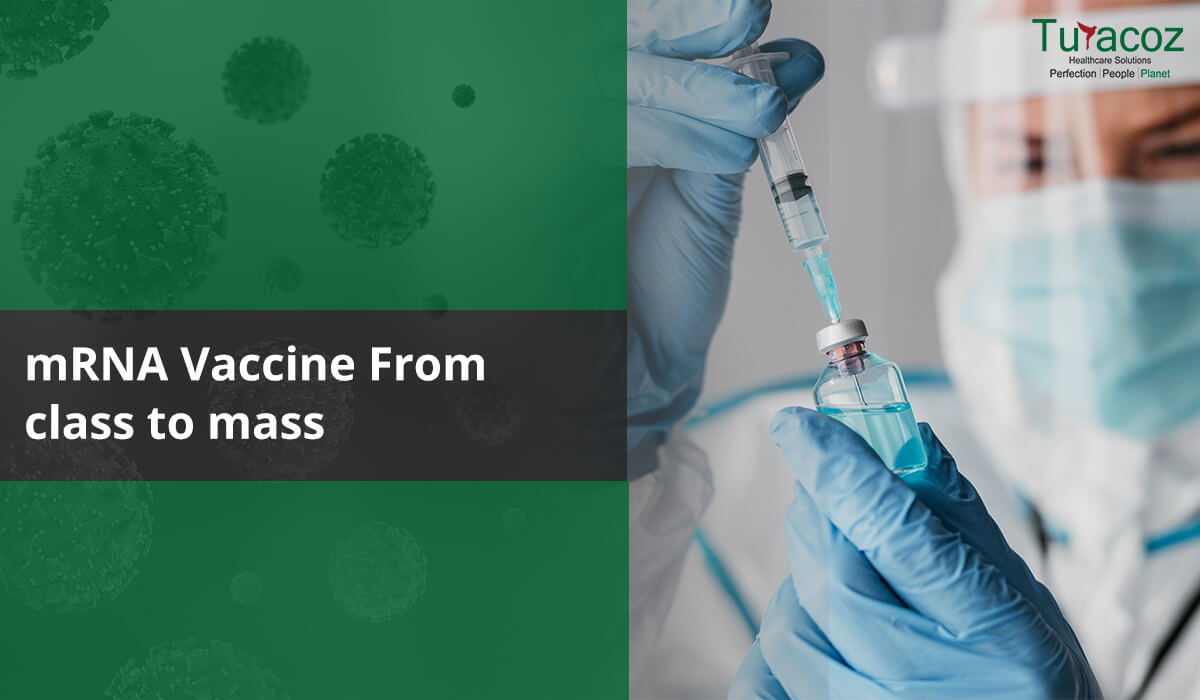
Coronavirus disease 2019(COVID-19) is giving the world a rollercoaster ride with shoot-up in infection cases globally. With more than 14 crores of infection cases worldwide in 16 months, the world is looking up to a miracle to stop this pandemic. One way of controlling the spread is to get vaccinated. More than 98 crores vaccines have been administered globally till the end of April 2021 and it is still making way to reach people in every corner of the world. India is in its second wave of COVID-19 infection with a tremendous rise in corona infection cases compared to the first wave of infection. But a positive side of this is, the largest vaccination drive of the world is under progress in India and giving positive hope for Indians to overcome this situation.

Class I devices are the lowest risk medical devices. However, the manufacturers of these devices also need to act immediately to comply with the new European Union (EU) Medical Devices Regulation (MDR); otherwise, they risk being unable to place their devices on the EU market after May 26, 2021.

Sleep is an important part of our day-to-day life and everybody long for a good night’s sleep after a day’s work. But do you know why you sleep at night and are awake during daytime? Do you know we have a biological clock inside us? Do you know why some people fall asleep while talking or even while driving? If you want to know the answers to these questions, please read on.

53 / 100 Powered by Rank Math SEO SEO Score What is pain? Pain can be a displeasing and uncomfortable

70 / 100 Powered by Rank Math SEO SEO Score Working from home comes with its own set of pros

69 / 100 Powered by Rank Math SEO SEO Score Background According to European Commission a web-based portal EUDAMED is

53 / 100 Powered by Rank Math SEO SEO Score As the Medical Device Regulation (MDR) deadlines are approaching for

The COVID-19 pandemic has overwhelmed our healthcare delivery systems and has effected the delivery of medical care across the spectrum. Medical device companies have also been hit by the crisis
as they struggle to make decisions regarding supply chains, and regulatory obligations in the midst of uncertainty.
Please find our blog discussing the impact of the pandemic on medical device regulations.

Partner with Turacoz to bring science to life through strategic and evidence-based communication.